Therefore, the sheer number of chlorine electrons does not affect the energy change seen by any given one. “How much energy is transferred per coulomb of charge?” While three times as many chlorine atoms must be reduced than aluminum, the stability imparted by that transformation (the “willingness”), is the same for each chlorine electron. The reason why is simple: remember that voltage is a ratio. Some may be wondering “if the reduction half-reaction is multiplied by three for the net reaction, why isn’t the potential multiplied by three?” Reduction potential is one of the few places in chemistry that stoichiometry doesn’t apply. Therefore, the opposite voltage is used, so. However, this is the OXIDATION of aluminum, NOT the reduction. įrom the table, the reduction of aluminum has a voltage of -1.66 volts. Here’s an example putting this all together:įrom the table, this half-reaction has a potential of +1.36 volts. , or the overall potential for the reaction, is simply the sum of and. denotes reduction potential and denotes oxidation. 
The notation for voltage in this context is (“e-naught”). Reduction potentials of some of the most common redox reagents Calculating the potential Because the value never changes, tables of standard reduction potentials are an electrochemist’s best friend. The potential is static for a given half-reaction. In the same way, summing the reduction and oxidation potentials gives the potential for the overall reaction. Summing the oxidation and reduction half-reactions gives the overall redox reaction equation. Conversely, the oxidation half-reaction shows the opposite of an electron being accepted, so it will be represented by the negative of the reduction potential. The more positive the reduction potential, the more favored a reduction is.īecause the reduction half-reaction is the process by which an atom/ion accepts an electron, the favorability of that half-reaction can be represented by the reduction potential. The reduction potential is different than electronegativity and electron affinity, but is related to both. This is called the standard reduction potential, and it’s measured in volts (J/C). Every chemical species, including atoms and ions, has a certain “willingness” to accept electrons. It represents the energy in Joules transferred by one Coulomb of charge. Potential, also known as voltage, is a very important concept in electrochemistry. Some elements, like vanadium, can even progress through multiple oxidation states as a redox reaction progresses. All redox reactions will have one species being oxidized and one species reduced. In this reaction, iron is reduced from an oxidation state of +3 to 0, while carbon is oxidized from an oxidation state of +2 to +4. In reactions that are not redox reactions, like acid-base reactions, the oxidation states of the elements do not change.Ī simple example of an oxidation reaction is the reaction of iron (II) oxide with carbon monoxide as follows: In a redox reaction, a simple mnemonic to remember the direction of the transfer of electrons is “Oil Rig,” which stands for “Oxidation is losing, reduction is gaining.” This refers to the fact that in oxidation, a species loses electrons while in reduction, the species gains electrons. 
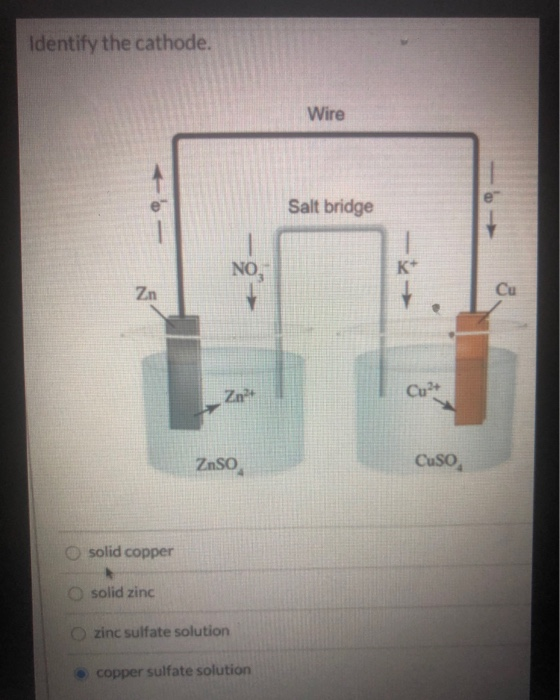
Redox reactions happen when a substance called an oxidizing agent oxidizes another substance by taking electrons and being reduced. In an electrochemical process, electrons flow from one substance to another substance in what is known as an oxidation-reduction (redox) reaction. Voltaic cells produce electricity, while electrolytic cells use a power source to drive a reaction forward. There are two types of electrochemical cells – a voltaic cell, also called a galvanic cell, and an electrolytic cell. You will also learn how to determine what half-reactions occur at which electrode and use these standard half-reaction potentials to calculate cell potential. In this tutorial on galvanic cells, aka voltaic cells, you will learn the basics of redox reactions and how to apply this information to electrochemical cells.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
